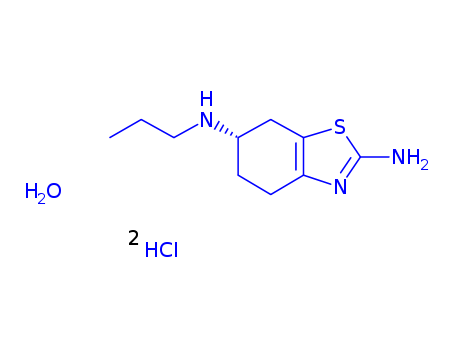
191217-81-9
- Product Name:Pramipexole
- Molecular Formula:C10H21Cl2N3OS
- Purity:99%
- Molecular Weight:302.268
Product Details
pd_meltingpoint:290 °C(dec.)
Appearance:gray white crystalline powder
Good supplier Hot Sale Top quality 191217-81-9 Pramipexole
- Molecular Formula:C10H17N3S*2ClH*H2O
- Molecular Weight:302.268
- Appearance/Colour:gray white crystalline powder
- Vapor Pressure:9.93E-11mmHg at 25°C
- Melting Point:290 °C(dec.)
- Boiling Point:378 °C at 760 mmHg
- Flash Point:182.4 °C
- PSA:88.41000
- LogP:4.09400
Pramipexole 191217-81-9 Usage
Pramipexole is a dopamine agonist that is well-tolerated and effective at treating Parkinson’s disease symptoms. Pramipexole was first manufactured by Pharmacia and Upjohn in July 1997 under the United States brand names of Mirapex and Mirapex ER. Pramipexole is classified as a nonergoline aminobenzothiazole compound that selectively agonizes the dopamine D2-like receptor subfamily, which includes the D2, D3, and D4 receptor subtypes. Pramipexole is a unique compound in its therapeutic potential because it has D3-preferring properties. The D3 receptor target has implications in both motor and psychiatric symptoms of Parkinson’s disease, restless leg syndrome, and bipolar and unipolar depression. Currently, pramipexole is approved to treat signs and symptoms of idiopathic Parkinson’s disease and moderate to severe symptoms of primary restless leg syndrome. Parkinson’s disease is characterized by tremor, bradykinesia, rigidity, gait disorders, and a disturbance of posture due to a decrease in dopamine stores in the substantia nigra with the consequent presence of Lewy bodies. Restless leg syndrome is a neurologic sensorimotor disorder characterized by a compelling urge to move the body/limb to relieve this uncomfortable sensation.
InChI:InChI=1/C10H17N3S.2ClH.H2O/c1-2-5-12-7-3-4-8-9(6-7)14-10(11)13-8;;;/h7,12H,2-6H2,1H3,(H2,11,13);2*1H;1H2
Pramipexole 191217-81-9 Relevant articles
Precise Dosing of Pramipexole for Low-Dosed Filament Production by Hot Melt Extrusion Applying Various Feeding Methods
Rebecca Chamberlain ,Hellen Windolf ,Simon Geissler ,Julian Quodbach and Jörg Breitkreutz
Pharmaceutics 2022, 14(1), 216
It was found that deviation from target pramipexole concentration occurred due to degradation products in bPMMA formulations. Additionally, material temperature above 120 °C led to the formation of the anhydrous form of pramipexole within the extruded filaments and need to be considered in the calculation of the recovered API.
Direct and sensitive electrochemical evaluation of pramipexole using graphitic carbon nitride (gCN) sensor
Yogesh M. Shanbhag ,Mahesh M. Shanbhag ,Shweta J. Malode ,S. Dhanalakshmi ,Kunal Mondal andNagaraj P. Shetti
Biosensors 2022, 12(8), 552
Pramipexole (PMXL) belongs to the benzothiazole class of aromatic compounds and is used in treating Parkinson’s disease; however, overdosage leads to some abnormal effects that could trigger severe side effects.
Efficacy of pramipexole on quality of life in patients with Parkinson’s disease: a systematic review and meta-analysis
Tao Li, Shuang Zou, Zijuan Zhang, Meiruo Liu & Zhanhua Liang
BMC Neurology, Volume 22, article number 320, (2022)
A systematic literature search of PubMed, Embase and the Cochrane Library was performed from inception to 30 April 2022 to identify randomised, placebo-controlled trials of patients with idiopathic PD receiving pramipexole, who reported a change from baseline in their QoL as measured by the 39-item Parkinson’s Disease Questionnaire (PDQ-39). Risk of bias was independently assessed by two reviewers using the Cochrane Collaboration’s tool for bias assessment.
191217-81-9 Upstream products
-
104632-26-0
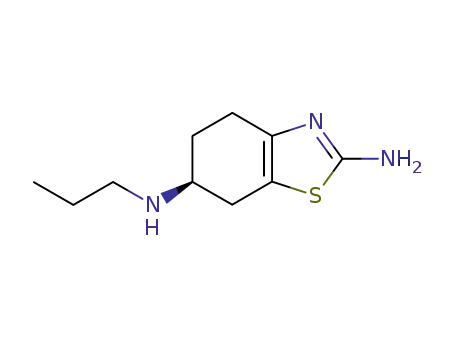
pramipexole
-
943319-02-6
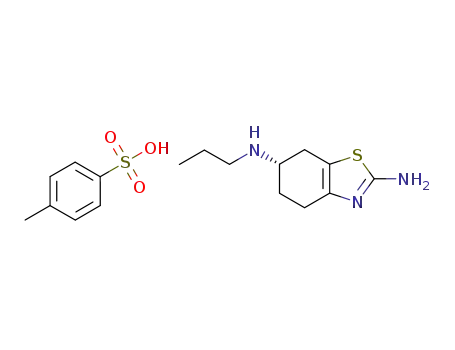
2-amino-6-propylamino-4,5,6,7-tetrahydrobenzothiazole p-toluenesulfonic acid
-
104632-25-9
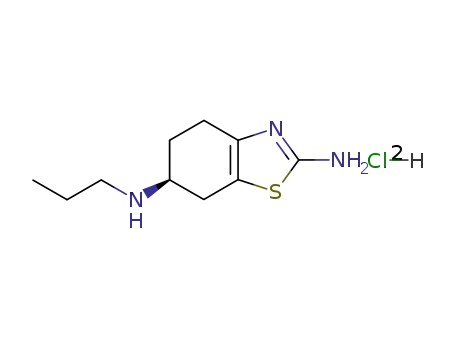
pramipexole dihydrochloride
-
106006-84-2
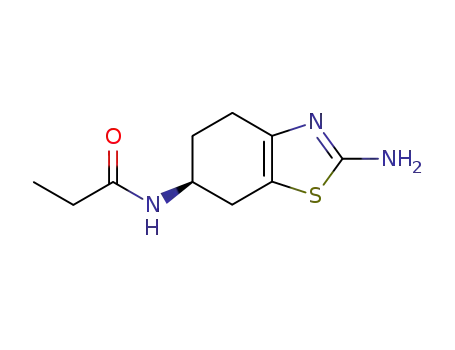
(S)?2?amino?6?propionamido?4,5,6,7?tetrahydrobenzothiazole
Relevant Products
-
Acotiamide HCl
CAS:773092-05-0
-
Tianeptine sodium salt
CAS:30123-17-2
-
99%Fenofibrate with high quality
CAS:49562-28-9