171596-29-5
- Product Name:Tadalafil
- Molecular Formula:C22H19N3O4
- Purity:99%
- Molecular Weight:
Product Details
Appearance:white powder
Throughput:10000|Kilogram|Year
pd_productuse: Male Sex Hormones
Delivery Time:3days
Shanghai Upbio Tech Co.,Ltd is a good supplier of Tadalafil . You can buy high purity Tadalafil 171596-29-5 powder In Medicine with the best price here.
High Quality Raw materials of health products Tadalafil 171596-29-5 In Medicine Good Supplier
- Molecular Formula:C22H19N3O4
- Molecular Weight:389.411
- Appearance/Colour:white to off-white cyrstalline solid
- Vapor Pressure:5.29E-13mmHg at 25°C
- Melting Point:298-300 °C
- Refractive Index:1.705
- Boiling Point:679.1 °C at 760 mmHg
- PKA:16.68±0.40(Predicted)
- Flash Point:364.5 °C
- PSA:74.87000
- Density:1.51 g/cm3
- LogP:2.08710
Tadalafil 171596-29-5 Usage
Tadalafil is a PDE5 inhibitor that acts as a vasodilator through the NO-cyclic guanosine monophosphate (cGMP) pathway and is prescribed for erectile dysfunction and PPHN. Tadalafil reportedly improves vascular endothelial dysfunction and suppresses inflammation.
Tadalafil is a PDE5 inhibitor with several promising features:
(1) it has a longer half-life and shows a more rapid onset of action than does sildenafil ;
(2) food intake has negligible effects on its bioavailability;
(3) it is particularly selective for the PDE5 enzyme and shows fewer side effects than sildenafil.
Tadalafil has been administered to vasodilate the pulmonary artery and improve pulmonary arterial hypertension (PAH).
Tadalafil is currently only available in Germany via a prescription.
InChI:InChI=1/C22H23N3O3/c1-24-8-9-25-17(22(24)26)11-15-14-4-2-3-5-16(14)23-20(15)21(25)13-6-7-18-19(10-13)28-12-27-18/h2-7,10,15,17,20-21,23H,8-9,11-12H2,1H3/t15?,17-,20?,21-/m1/s1
171596-29-5 Relevant articles
Tadalafil for Treatment of Fetal Growth Restriction: A Review of Experimental and Clinical Studies
S Maki, S Takakura, M Tsuji, S Magawa, Y Tamaishi
Biomedicines 2024, 12(4), 804;
Tadalafil is a promising therapeutic agent for FGR that can improve placental function in terms of uteroplacental perfusion and nutritional exchange. Research on tadalafil has gradually advanced, and to date no other therapeutic argent has received greater research interest for the treatment of FGR, which makes it a practical option. Although the present study focused on treatment for FGR, in the future we will also investigate tadalafil from the perspective of a preventive drug for placental insufficiency. A placebo-controlled, randomized controlled trial, TADAFER IIb, is being conducted; case registration is ongoing, and we await the results.
Evidence for benefits and risks of tadalafil as a non-prescription medicine: review and evaluation using the Group Delphi technique to achieve consensus amongst clinical experts
K Miller, U May, WD Beecken
Front. Pharmacol., 09 October 2023
Based on this thorough and systematic assessment of the evidence for the incremental benefits and risks of making tadalafil available without prescription in Germany, the consensus amongst clinical experts was that the incremental risks would be small. They would either be unlikely to occur or, if more likely to occur, would have little clinical impact. There was high level agreement that the risks are sufficiently manageable, so even if there were differing opinions regarding the magnitude of benefits, the average risk evaluation appeared significantly lower than the potential benefits.
171596-29-5 Upstream products
-
74-89-5
methylamine
-
171489-59-1
methyl (1R,3R)-1-(3,4-methylenedioxyphenyl)-2-chloroacetyl-2,3,4,9-tetrahydro-9H-pyrido[3,4-b]indol-3-carboxylate
-
749864-18-4
1-benzo[1,3]dioxol-5-yl-2-{[(9H-fluoren-9-ylmethoxycarbonyl)-methyl-amino]-acetyl}-2,3,4,9-tetrahydro-1H-β-carboline-3-carboxylic acid methyl ester
-
951661-81-7
(1R,3R)-1-(benzo[d][1,3]dioxol-5-yl)-2-(2-chloroacetyl)-N-methyl-2,3,4,9-tetrahydro-1H-pyrido[3,4-b]indole-3-carboxamide
171596-29-5 Downstream products
-
171596-27-3
(6R,12aS)-2,3,6,7,12,12a-hexahydro-2-methyl-6-(3,4-methylenedioxyphenyl)-pyrazino[2',1':6,1]pyrido[3,4-b]indole-1,4-dione
-
1220393-12-3
3-((2-(benzo[d][1,3]dioxole-5-carbonyl)-1H-indol-3-yl)methyl)-1-methylpiperazine-2,5-dione
-
1242099-13-3
C10H12O3*C22H19N3O4
-
1242099-11-1
C8H8O3*C22H19N3O4
Relevant Products
-
Cyclopropane, isocyanato- 4747-72-2
CAS:4747-72-2
-
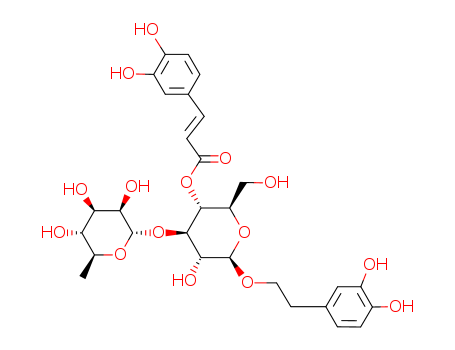
Acteoside/Verbascoside
CAS:61276-17-3
-
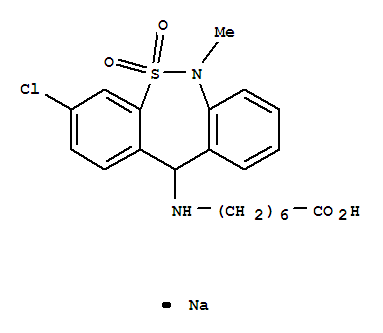
Tianeptine sodium salt
CAS:30123-17-2